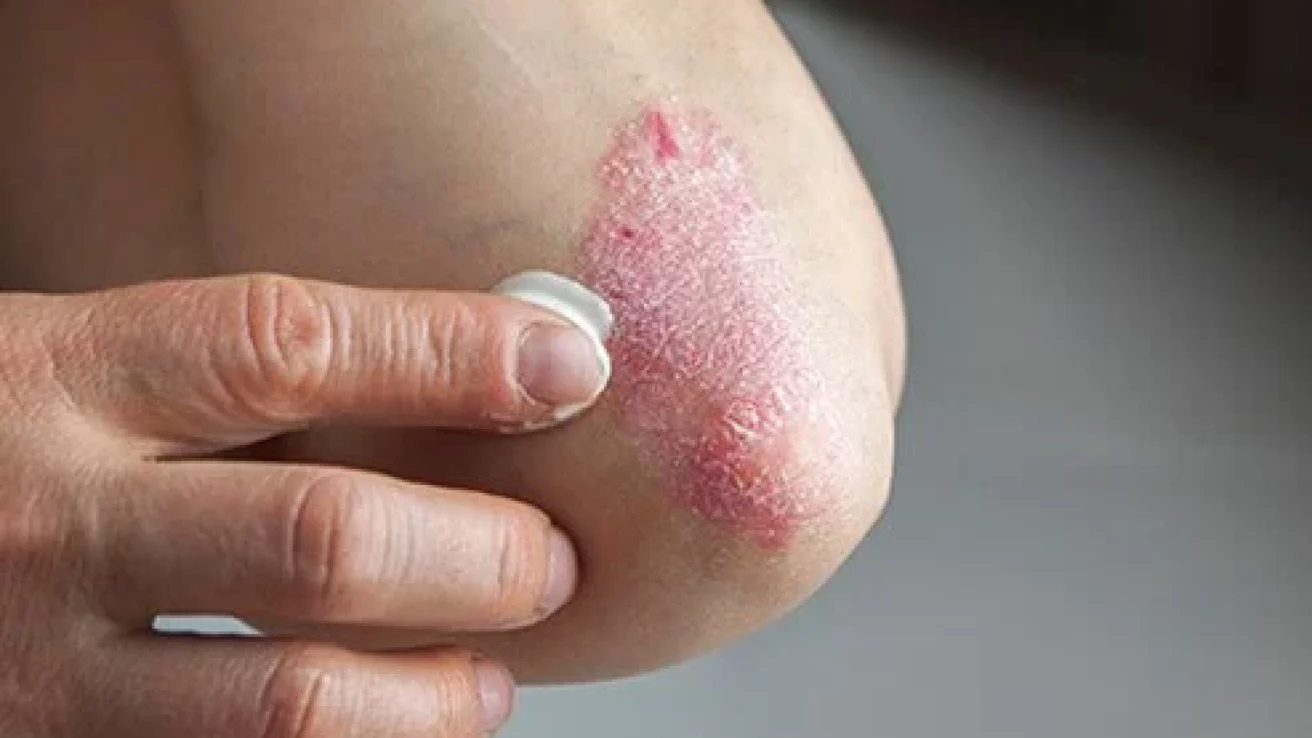
Difamilast ointment is superior to vehicle in terms of severity index score and overall eczema area in patients with AD, according to a recent trial, which reported only mild to moderate adverse effects of difamilast.
Atopic dermatitis (AD) is an inflammatory skin condition that frequently occurs in children. AD has a relapsing-remitting nature and is identified as a lifelong chronic inflammatory skin condition. The mainstay of AD treatment is avoidance of exposure to triggers, topical anti-inflammatory agents, and topical therapies, including emollients and moisturizers. Difamilast is described as a selective phosphodiesterase 4 (PDE4) inhibitor. PDE4 is associated with the production of cytokines in inflammatory conditions, including AD. The study, published in the Journal of the American Academy of Dermatology, was a phase 3 randomized trial that aimed to recognize the superiority of 1% difamilast ointment over its vehicle in Japanese AD patients.
The primary endpoint of the study was the investigator global assessment (IGA) score at week four. Of the 414 participants included in this trial, 364 were administered a minimum of one dose of the study medication. Three hundred of these participants completed the trial. The remaining participants discontinued owing to consent withdrawal and the onset of adverse events.
The IGA score in the 4th week was significantly higher in patients administered 1% difamilast ointment as compared to patients administered the vehicle, (38.46% vs 12.64%, respectively, P < .0001). Significant improvements were also observed in the severity index score and overall eczema area with 1% difamilast ointment compared to the vehicle. Moreover, adverse events were mild or moderate in intensity and occurred less frequently with 1% difamilast ointment. The study concluded that difamilast is superior to the vehicle and is safe for individuals with AD.
References
Saeki, H., Ito, K., Yokota, D., & Tsubouchi, H. (2022). Difamilast ointment in adult patients with atopic dermatitis: A phase 3 randomized, double-blind, vehicle-controlled trial. J Am Acad Dermatol, 86(3), 607-614. doi:10.1016/j.jaad.2021.10.027